 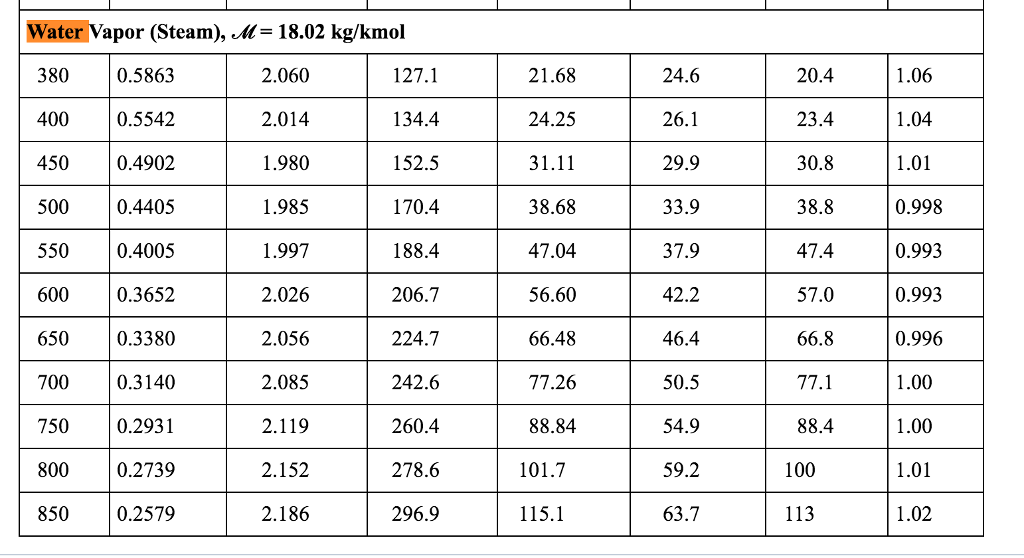
Symbols, abbreviations, or full names for units of length,Īrea, mass, pressure, and other types. You can find metric conversion tables for SI units, as wellĪs English units, currency, and other data. It is the pressure resulting from a force of one pound-force applied to an area of one square inch.Ĭonversion calculator for all types of measurement units. 
The pound per square inch or, more accurately, pound-force per square inch (symbol: psi or lbf/in² or lbf/in²) is a unit of pressure or of stress based on avoirdupois units. Psi to foot of head, or enter any two units below: Enter two units to convert From: Try a couple of practice questions yourself and check the video answers to see how you did.You can do the reverse unit conversion from Step 2 (really step 1): We first need to change the feet of head to psi. If the liquid is not water (examples: paint, chocolate syrup or gasoline), the liquid’s specific gravity must be factored into the formula. Let’s put all the main players when dealing with pressure into a table with the starting point being one pound per square inch.Īs we have done in the previous parts of the chapter it’s time to go through some examples working between these numbers. This constant converts a foot of head of water into pressure: Head in feet of water divided by 2.31 equals pressure in psi, and pressure in psi times 2.31 equals head in feet. In any event a Torr is roughly equal to 133.32 Pascals. Standard atmospheric pressure is also known to be 14.7 psi. You might sometimes also hear it referred to as standard atmospheric pressure. One atmosphere is essentially the pressure at sea level exerted on all objects. It is now defined as 1/760 of an atmosphere where one atmosphere is defined as 101.325 kPa. Torr: Was originally meant to be equal to 1 mmHg but it’s not quite that anymore. 
Let’s throw in another few examples of ways pressure can be expressed.īar: Approximately the average pressure at sea level. 2.04 inches of mercury is equal to 1 pound per square inch. We would be required to measure vacuum for such things as vacuum pumps in medical facilities or to measure vacuum in refrigeration systems. Inch of mercury (Hg): This one we might actually use as its common when measuring a vacuum. Inches of mercury (Hg) is another common way to measure pressure. If you are wondering if this will work for other liquids the answer is yes. As it turns out 27.72 inches is equal to 2.31 feet so if we had a column of water 27.72 inches (2.31 feet) we would have a pressure of one pound per square inch at the base of that column of water. The pressure at the base of that column of water would be 1 psi. One pound per square inch is equivalent to having a column of water 2.31 feet in height. The higher the column of water the greater the pressure. The idea is that having a column of water creates a pressure at the base of the column. Another couple of pressure measurements that are common in the world of trades are inches of water column and feet of head.īoth are in reference to a column of water. Those are not the only pressure measurements that we’ll deal with. One being the Pascal (Pa) (or kilopascal(kPa)) and the other being pounds per square inch (psi). Whatever the case we basically have our two measurements of force to work with. It is sometimes referred to as pounds-force or lbf. We can also include pounds in the idea of force where pounds can be defined as a unit of force as well. Newton: is the force needed to move (or accelerate) an object weighing one kilogram at one metre per second squared. If we want to get technical here is the definition of what a newton is. One Pascal is equal to a pressure of one Newton per metre square (n/m2) where a Newton is a measure of force. Now as pressure is defined as force per unit area we should define what a Pascal actually is. The most common metric version of pressure is known as the Pascal (Pa) and quite often we put Pascals into groups of 1000 and just like the metric system likes to do it’s called a kilopascal (kPa). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
